Ana Santos Rutschman at Saint Louis University Law School has authored an interesting short article titled, "Why Moderna Won't Share Rights to the COVID-19 Vaccine with the Government that Paid for Its Development" in The Conversation. The article basically outlines the U.S. government's technical and monetary contributions to the development of the mRNA technology and a dispute between Moderna and the U.S. government. The article is available, here. Moderna's stock has been falling overall, and I imagine this will not help as this is resolved. I've been worried about vaccine availability for some time, but I didn't realize so many would choose not to be vaccinated. It appears COVID-19 mutation will continue relatively unabated.
"Where money issues meet IP rights". This weblog looks at financial issues for intellectual property rights: securitisation and collateral, IP valuation for acquisition and balance sheet purposes, tax and R&D breaks, film and product finance, calculating quantum of damages--anything that happens where IP meets money.
Saturday, 20 November 2021
Friday, 12 November 2021
The TRIPS Trap Revisited
By Roya Ghafele, OxFirst
The World Trade Organization’s Trade-Related Aspects of Intellectual
Property Rights (TRIPS) Agreement forced much of the
developing world to adopt minimal standards for intellectual property (IP) protection.
The imposition of such standards has been subject to much criticism. NGOs and
advocates of international development have raised serious concerns about TRIPS,
TRIPS Plus (which expands existing obligations under TRIPS), and the increasing
use of bilateral trade agreements to expand IP protection.
Critics of such agreements cite the costs associated with
enacting IP legislation and that, due to global IP protection, imitation as a
development strategy has been ruled out. These concerns can neither be denied,
nor is it the aim of this article to dismantle them. They are certainly important problems that
deserve to be further studied.
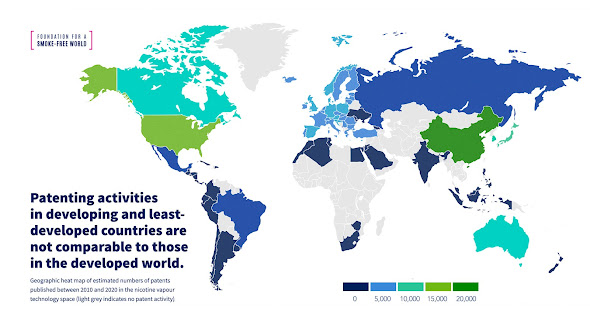
However, preliminary insights in the patent positions in
technologies relating to tobacco harm reduction suggests that out of 74 000
patents published on technologies aiming to reduce the harm of smoking, hardly
any cover the developing world.
With the exception of South Africa, the typical African
country has not seen any significant patent filings in this technology.
Figure 1: Indications of patents relating to vaping over
the last decade
There may certainly be conditions lying outside of the patent system that have discouraged tobacco companies from creating IP in these countries. However, the significant difficulties developing countries may have faced with implementing the patent system, may also have discouraged patenting activities in the developing countries.
Many developing countries have struggled setting up
functioning patent offices, finding qualified staff, training judges in IP and
developing local IP professionals. in some countries patent offices may have
been established without the necessary infrastructure or human capacity to
successfully examine patent applications. IP legislation has been enacted
without investing in the necessary infrastructure to turn legal theory into
legal practice. None of this may have been undertaken in bad faith or to
wilfully breach a commitment to international trade.
Rather, the sad reality is that in many developing
countries there is very little understanding about IP and its economic impact. Even
at the European level, the qualifying exam for European patent attorneys is
more successfully passed by Brits or Germans, rather than their colleagues from
less-privileged countries. Greece, for example, has up until now not succeeded
in developing patent attorneys and the job is undertaken by lawyers.
Lack of know-how and know-why, are probably the primary
reasons many developing countries are still grappling with the IP system. Leading
IP economies have often been less than understanding about the constraints
facing developing nations. The Office of the US Trade Representative for
example releases annually a special report on IP and review of notorious
markets for counterfeiting and piracy.
International development aid for IP has been rather scattered.
The World Intellectual Property Organization (WIPO) followed the United States
Agency for International Development (USAID) offer the most important support
for IP-related development assistance. However, at the international level technical
assistance can often be neither coordinated nor continuous. With the exception
of WIPO, such assistance may also have a tendency to reflect the specific
requirements of the bilateral donor country, rather than focus on the larger
needs of a developing country or an entire region.
All of this has led to the current sorry state of global IP
law. While some nations are at the forefront of defining the technology law of
the future, others are still confronted with a situation where ordinary people
have probably not even heard of intellectual property.
The initial analysis in the tobacco space was a harsh
awakening as to how at least one sector of the economy assesses the value
proposition of developing countries’ IP systems. Tobacco companies are waging
trans-jurisdictional patent battles over next-generation tobacco products, but
at the same time their focus on developing country IP is minimal.
This is at odds with the market reality. Over 85% of
smokers live in developing countries, yet the patent focus remains on developed
nations. The market potential for tobacco harm-reducing products in developing
nations is substantial, if these products were to be made available globally,
rather than to consumers in developed countries only. This would not only
expand business opportunities for the tobacco industry, but would also offer people
around the globe less harmful ways to consume tobacco. A potential “win-win”
situation is being hampered by a deplorable implementation of the patent system
in many countries.
I do not want to be misunderstood as a hard-core advocate
of the patent system. However, the initial patent landscape analysis made me
wonder if in the 21st century any nation can truly afford to stay outside of
that system. The reality is that, doing so, translates into marginalization. This
segregation may be so pronounced that companies may not be motivated to invest
in patents in these countries. Usually, this also means that they have no
intention of selling patented products in these countries, leading to restricted
consumer choice and limited take-off of technology. Whether this is indeed a
better development choice should be a subject of further discussion.
Key words: smoking, tobacco hard reduction, patent protection, low- and middle-income countries
Disclosure of Conflict of Interest: OxFirst has been compensated by the Foundation for a Smoke-Free World, the views are
the author’s own. The preliminary patent landscape report is available here and here.
Saturday, 6 November 2021
U.S. DOJ Settles Generic Drug Price Fixing Cases -- Over $400 million
The U.S. Department of Justice has settled several lawsuits concerning essentially of illegal price fixing between companies offering generic drugs. This is a continuation of the problems facing drug pricing and the generic market. Government production of generics may be the future. The U.S. Department of Justice press release states, in part:
Three generic pharmaceutical manufacturers, Taro
Pharmaceuticals USA, Inc., Sandoz Inc. and Apotex Corporation, have agreed to
pay a total of $447.2 million to resolve alleged violations of the False Claims
Act arising from conspiracies to fix the price of various generic drugs. These
conspiracies allegedly resulted in higher drug prices for federal health care
programs and beneficiaries according to the Justice Department.
The government alleges that between 2013 and 2015, all three
companies paid and received compensation prohibited by the Anti-Kickback
Statute through arrangements on price, supply and allocation of customers with
other pharmaceutical manufacturers for certain generic drugs manufactured by
the companies.
Taro Pharmaceuticals USA, Inc., headquartered in New York,
has agreed to pay $213.2 million. The Taro drugs allegedly implicated in this
scheme address a wide variety of health conditions, and include etodolac, a
nonsteroidal anti-inflammatory drug used to treat pain and arthritis, and
nystatin-triamcinolone cream and ointment, a combination of an antifungal
medicine and steroid used to treat certain kinds of skin infections.
Sandoz Inc., headquartered in New Jersey, has agreed to pay
$185 million. The Sandoz drugs at issue include benazepril HCTZ, used to treat
hypertension, and clobetasol, a corticosteroid used to treat skin conditions.
Apotex Corporation, headquartered in Florida, has agreed to
pay $49 million in connection with its sale of pravastatin, a drug used to
treat high cholesterol and triglyceride levels.
“Illegal collaboration on the price or supply of drugs
increases costs both to federal health care programs and beneficiaries,” said
Acting Assistant Attorney General Brian M. Boynton of the Justice Department’s
Civil Division. “The department will use every tool at its disposal to prevent
such conduct and to protect these taxpayer-funded programs.”
“These civil settlements are another achievement in my
office’s efforts to hold generic drug companies accountable for the consequences
arising from price-fixing schemes, including the harm to federal health care
programs,” said Acting U.S. Attorney Jennifer Arbittier Williams for the
Eastern District of Pennsylvania. “We will continue to aggressively pursue
these violations of the Anti-Kickback Statute and the False Claims Act and
obtain significant recoveries.”
“Conspiring to raise prices on generic medications is illegal
and could prevent patients from being able to afford their needed prescription
drugs. Americans have the right to purchase generic drugs set by fair and open
competition, not collusion,” said Special Agent in Charge Maureen R. Dixon of
the Philadelphia Regional Office of the Inspector General, Department of Health
and Human Services (HHS-OIG). “HHS-OIG along with our law enforcement partners
will continue to investigate allegations of companies engaging in actions that
put the public and the Medicare program at risk.”
In connection with its settlement agreement, each company
also entered a five-year corporate integrity agreement (CIA) with OIG. The CIAs
include unique internal monitoring and price transparency provisions. They also
require the companies to implement compliance measures including risk
assessment programs, executive recoupment provisions and compliance-related
certifications from company executives and board members. . . .
The Anti-Kickback Statute prohibits companies from receiving
or making payments in return for arranging the sale or purchase of items such
as drugs for which payment may be made by a federal health care program. These
provisions are designed to ensure that the supply and price of health care
items are not compromised by improper financial incentives. These settlements
reflect the important role of the False Claims Act to ensure that the United
States is fully compensated when it is the victim of kickbacks paid to further
anticompetitive conduct.
All three companies previously entered into deferred
prosecution agreements with the Antitrust Division to resolve related criminal
charges. Taro paid a criminal penalty of $205.6 million and admitted to
conspiring with two other generic drug companies to fix prices on certain
generic drugs. Sandoz paid a criminal penalty of $195 million and admitted to
conspiring with four other generic drug companies to fix prices on certain
generic drugs. Apotex paid a criminal penalty of $24.1 million and admitted to
conspiring to increase and maintain the price on pravastatin. The civil
settlement payments announced today are in addition to the criminal penalties
paid by the companies.
The press release is available,
here.
Thursday, 4 November 2021
Morrison and Foerster Sponsors Webinar titled, "In a Founders Market, Strategic Capital Thrives."
The law firm Morrison & Foerster (a very famous law firm based in California known as MoFo) is sponsoring a free webinar on the “buyer’s market” for capital titled, “In a Founders Market, Strategic Capital Thrives.” The webinar is scheduled for “Tuesday, November 16, 2021[,] 9:00 a.m. – 10.00 a.m. PT[,] . . . 5:00 p.m. – 6:00 p.m. GMT.”
Additional details are below:
|
2021
is turning out to be a record-setting year for investment in VC-funded
startups and private companies. This vast global liquidity has caught the
eyes of non-traditional investors including sovereign wealth funds and
limited partners, both of whom are looking to boost returns with direct investments. This makes it a
buyer’s market for startups, and the best entrepreneurs have access to what
seems like unlimited capital.
Panelists Include:
|
RSVP is available, here.
Wednesday, 3 November 2021
U.S. FDA Sends Letter to Push USPTO Concerning Drug Patents and Access
The U.S. Federal Food and Drug Administration (FDA) recently sent a letter to the U.S. Patent and Trademark Office concerning the FDA’s concerns regarding pharmaceutical patents and their impact on innovation and access. Some of the concerns include the use of continuations to build patent thickets to raise litigation costs as well as resulting in possible delays of generic entry; evergreening practices; and product-hopping. Notably, the FDA is generally interested in increasing communication and collaboration to address those issues, including offering expertise, collecting additional information regarding IPRs and other post-grant procedures as well as inquiring whether examiners need more time to review patent applications. While the Trump Administration also had concerns regarding drug pricing, President Biden’s recent Executive Order concerning competition is the impetus for this letter’s push for increased collaboration.
U.S. Federal Trade Commission Releases New Safeguards Rule for Non-Banking Financial Institutions
The Federal Trade Commission (FTC) in the United States has changed the regulations concerning the Safeguards Rule relating to cybersecurity standards for non-banking financial institutions. Essentially, the new Safeguards Rule contains additional specificity regarding what is required to comply with the contextual administrative, physical and technical standards for a compliant information security program. The new Safeguards Rule will be effective a year from publication in the Federal Register. Notably, the new Safeguards Rule contains significant new definitions. The FTC press release states, in relevant part:
The FTC’s updated Safeguards Rule requires non-banking
financial institutions, such as mortgage brokers, motor vehicle dealers, and
payday lenders, to develop, implement, and maintain a comprehensive security
system to keep their customers’ information safe.
“Financial institutions and other entities that collect
sensitive consumer data have a responsibility to protect it,” said Samuel
Levine, Director of the FTC’s Bureau of Consumer Protection. “The updates
adopted by the Commission to the Safeguards Rule detail common-sense steps that
these institutions must implement to protect consumer data from cyberattacks
and other threats.”
The changes adopted by the Commission to the Safeguards
Rule include more specific criteria for what safeguards financial
institutions must implement as part of their information security program such
as limiting who can access consumer data and using encryption to secure the
data. Under the updated Safeguards Rule, institutions must also explain their
information sharing practices, specifically the administrative, technical, and
physical safeguards the financial institutions use to access, collect,
distribute, process, protect, store, use, transmit, dispose of, or otherwise
handle customers’ secure information. In addition, financial institutions
will be required to designate a single qualified individual to oversee their
information security program and report periodically to an organization’s board
of directors, or a senior officer in charge of information security.
The Safeguards Rule was mandated by Congress under the 1999
Gramm-Leach-Bliley Act. Today’s updates are the result of years of public
input. In 2019, the FTC sought
comment on proposed changes to the Safeguards Rule and, in 2020
held a
public workshop on the Safeguards Rule.
In addition to the updates, the FTC is seeking comment on
whether to make an additional change to the Safeguards Rule to require
financial institutions to report certain data breaches and other security
events to the Commission. The FTC is issuing a supplemental notice of proposed rulemaking,
which will be published in the Federal Register shortly. The public will have
60 days after the notice is published in the Federal Register to submit a
comment.
The new Safeguards Rule is available, here. Notably, there is legislation before the U.S. Congress to massively increase the budget of the FTC to deal, in part, with privacy and cybersecurity issues.